The demand for valuable metals needed in batteries is expected to increase significantly in the coming decades with the growth of clean energy technologies. However, the supply of these battery metals is not unlimited, and current strategies to collect them from their natural reserves are resource and pollution-intensive and unsustainable in the long term. The best place to source them may be by recycling spent batteries.
A team of researchers from Rice University has developed a battery recycling method that can help recover over 98% of metal from various types of mixed battery waste. The new method also significantly reduces secondary waste streams from the contaminated, acidic leaching solutions and cuts the duration of the recycling process by almost 100-fold.
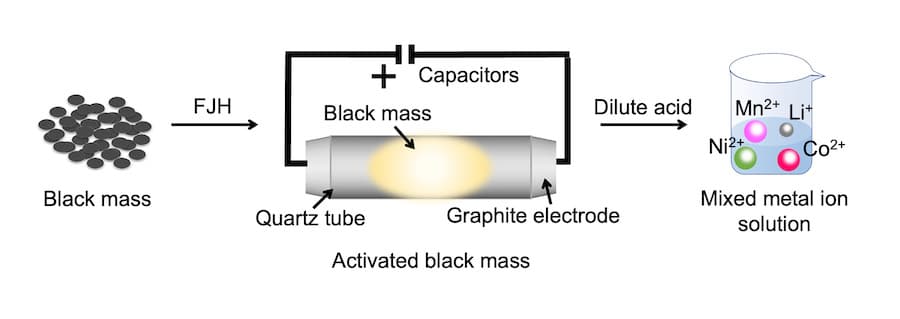
Researchers used their patented Joule-heating technique to bring the combined cathode and anode waste to temperatures above 2100 degrees Kelvin in seconds. This helped them remove the inert layer on battery metals and lower their oxidation state, making them soluble in low-concentration acid.
The new process takes less than 20 minutes to dissolve the same amounts compared to existing methods. This process can potentially reduce the cost of battery waste recycling through decreased energy, water, and acid consumption and lower carbon dioxide emissions.
“We developed a high-yield, low-cost method of reclaiming metals directly from ‘black mass’ – the combined cathode and anode waste the industry traditionally tries to recycle – that significantly reduces the environmental footprint of spent battery processing,” said Jinhang Chen, a co-lead author on a study.
Researchers say battery recycling is a very big deal, especially now, as many electric vehicles have been in use for about a decade, and many of their batteries are approaching their end of life now. Recycling spent batteries not only helps mitigate the environmental impacts of mining but also makes economic sense, as many kinds of lithium-ion batteries contain higher levels of metals, such as cobalt and nickel, than natural ores.
“Currently, 95% of batteries are not recycled because we don’t have the capacity to recycle them, even as waste from electronics is increasing at an annual rate of 9%,” said Rice chemist James Tour, whose lab previously developed a process for recycling battery anodes. “A lot of current battery recycling processes involve the use of very strong acids, and these tend to be messy, cumbersome processes.
“What we found is that if you ‘flash’ the black mass, then you can easily separate out the critical metals using only low-concentration hydrochloric acid. You could say the flash liberates the metals, so they dissolve more easily. We’re still using acid, but much less. That’s why the economics is so much better.”
The study includes a life-cycle assessment comparing the new process to different current methods of battery recycling.
“This study has the capacity to motivate the growth of battery waste management and contribute to the mass production of electric vehicles at a more competitive cost by lowering the cost of battery production,” Chen said in an official statement.
