High-quality, rechargeable batteries are a necessity to meet the world’s growing energy demands in a sustainable fashion, but not all are equal. Lithium-sulfur batteries are promising candidates for the next generation of these energy sources as they can store more energy than other rechargeable batteries. But the inherent instabilities are a key barrier to their widespread adoption.
Researchers in the Energy Materials and Surface Sciences Unit at the Okinawa Institute of Science and Technology Graduate University have proposed a promising solution to this problem by integrating a novel sponge material that allows a prototype lithium-sulfur battery to safely withstand hundreds of cycles.
“Lithium-sulfur batteries can store more energy than the lithium-ion batteries that are already commercially available,” said Dr. Hui Zhang, first author of this study. “To put this in numbers, an electric vehicle that runs on lithium-ion batteries can drive an average of 300km before it needs to be charged. With the improved energy storage provided by lithium-sulfur batteries, it should be possible to extend this to 500km.“
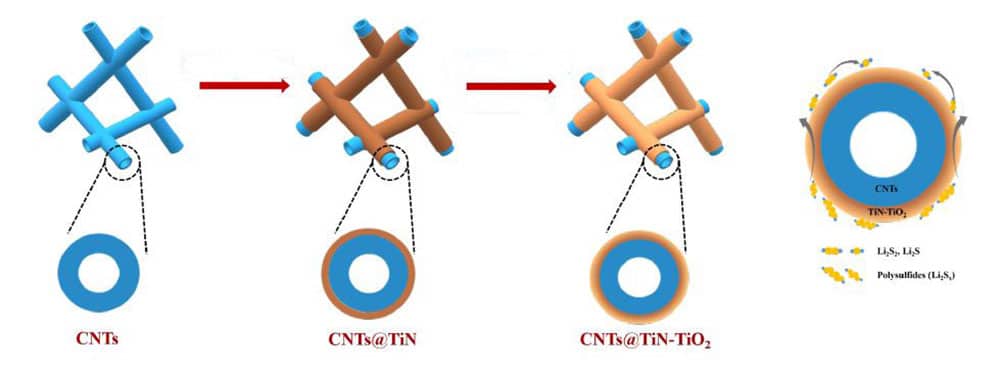
For lithium-sulfur batteries to become readily available, a common, dissolving issue during their construction needs to be overcome. During the construction of the battery, the sulfur will react with the lithium to form a product. A chemical reaction between lithium and sulfur first creates lithium polysulfide, which then can easily dissolve into polysulfides. If this happens, the polysulfides will impair the performance of the battery, resulting in its lifespan being greatly reduced.
To optimize the batteries, the lithium polysulfide needs to transform to the final product, either lithium persulfide (Li2S2) or lithium sulfide (Li2S), as quickly as possible. To do this, the researchers created a structure that could speed up the reaction process and absorb the unwanted polysulfides. They used a carbon nanotube framework (CNT) and coated it with titanium nitride (TiN) and titanium dioxide (TiO2), which offer some beneficial properties. The TiN acted as a material absorbing any unwanted polysulfides that were created in the process, whereas the TiO2 sped up the conversion from lithium polysulfides to the final products – Li2S2 or Li2S.
“Using these two materials, we developed a hybrid that is low cost and easy to apply,” said Dr. Luis Ono, second author of this study. “We found that it had an excellent ability to improve the battery performance.”
With the polysulfides being absorbed and the whole process being accelerated, the battery’s performance was greatly improved compared to versions without the hybrid sponge material. This translated to a shorter charging time, a longer life between charges thanks to high specific capacity, and a greater overall lifespan. To establish this, the researchers ran the battery for 200 cycles and found that its efficiency was almost the same.
“We will continue to further optimize the materials to improve the performance,” said Professor Yabing Qi, senior author of the study and head of the Energy Materials and Surface Sciences Unit at OIST. “There are a lot of brilliant minds working on lithium-sulfur batteries, and it’s a really promising and exciting technology.“
