Carbon capture and utilization technology, one of the methods for achieving net-zero CO2 emissions, has drawn attention as an innovative technology for reducing CO2 emissions. However, the high energy consumption required in the process of purification, pressurization, separation, and reuse of CO2 poses a challenge to the industrial application of these technologies in practice.
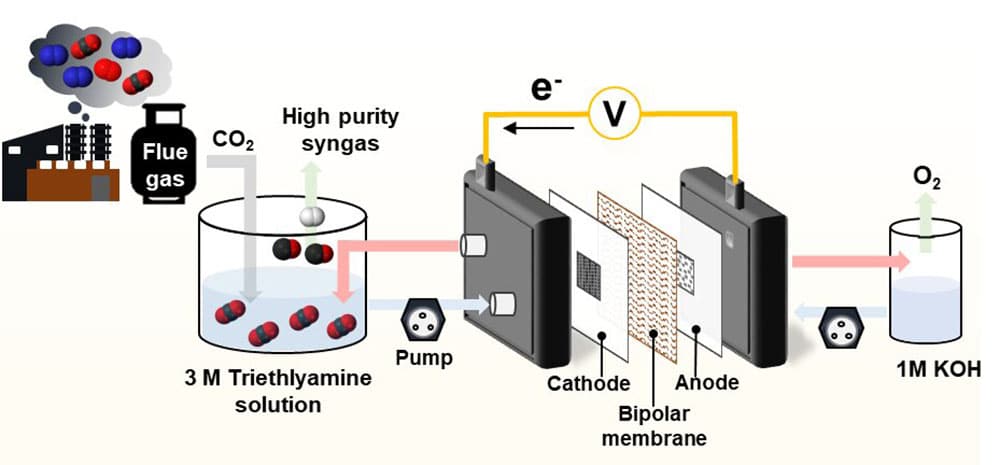
The research team at the Clean Energy Research Center, Korea Institute of Science and Technology (KIST), has developed a process for producing high-value-added synthesis gas (syngas) by direct electrochemical conversion of CO2 captured using a liquid absorbent.
The CO2 conversion process utilizes the CO2 captured in a liquid absorbent. This way, the conventional CCU (Carbon dioxide Capture Utilization) pathways with complex and energy-consuming processes of purification and pressurization of CO2 for pure gaseous CO2 production are no longer needed. This is why the proposed method outperforms the conventional CCUS technology, with superior cost-effectiveness and enhanced effect of reducing CO2 emissions.
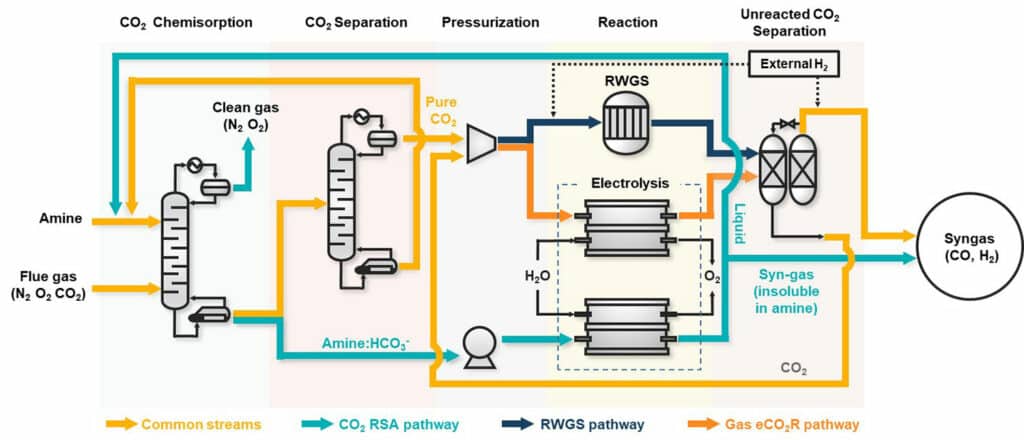
In addition, since unreacted CO2 is still captured in the liquid absorbent, there is no need for an additional separation process with syngas, a product from the pathway; another advantage is that the ratio of hydrogen to CO in the syngas can be more easily controlled.
The researchers were also able to maximize the efficiency of the direct CO2 conversion in the liquid phase by conducting experiments for selecting the best absorbent, optimizing the catalyst, designing an electrochemical reactor as well as testing long-term stability.
To examine the feasibility of commercialization of the developed process, the team also carried out simulation studies with numerical modeling of the industrial-scale process. It is estimated that the newly developed CO2 conversion process will be able to reduce production costs by 27.0% and CO2 emissions by 75.7% compared to the conventional CCUS technology.
Furthermore, the proposed technology demonstrated an equivalent level of competitive price when compared to the current market price of chemicals dominated by fossil fuel-based technologies. If the developed CO2 conversion process is applied to a major CO2 emission source such as a thermal power plant, the proposed technology is expected to be able to produce high-value chemicals such as ethylene at a low cost while reducing CO2.
“The significance of the proposed technology lies in that we have achieved technological progress in the efficient production of high-concentration syngas through the electrochemical process by utilizing captured CO2,” said Dr. Da Hye Won, a senior research scientist at KIST.
Dr. Ung Lee, the principal research scientist at KIST, commented, “We expect that the proposed technology will be applicable to a range of electrochemical conversion systems that utilize CO2, and we plan to move onto the next stage of continuous process demonstration and verification as well as technology transfer to business entities in the future.”
Journal reference:
- Kezia Megagita Gerby Langie, Kyungjae Tak, Changsoo Kim, Hee Won Lee, Kwangho Park, Dongjin Kim, Wonsang Jung, Chan Woo Lee, Hyung-Suk Oh, Dong Ki Lee, Jai Hyun Koh, Byoung Koun Min, Da Hye Won and Ung Lee. Toward economical application of carbon capture and utilization technology with near-zero carbon emission. Nature Communications 2022; DOI: 10.1038/s41467-022-35239-9
