Lithium-ion and even newer lithium-polymer batteries have a limited life and gradually lose their capacity to hold a charge. So recycling them is important in the long run in managing waste and bolstering reserves of critical materials.
Now, researchers at the Department of Energy’s SLAC National Accelerator Laboratory and Stanford University may have found a way to revitalize rechargeable lithium batteries, potentially boosting the range of electric vehicles and battery life in next-gen electronic devices.
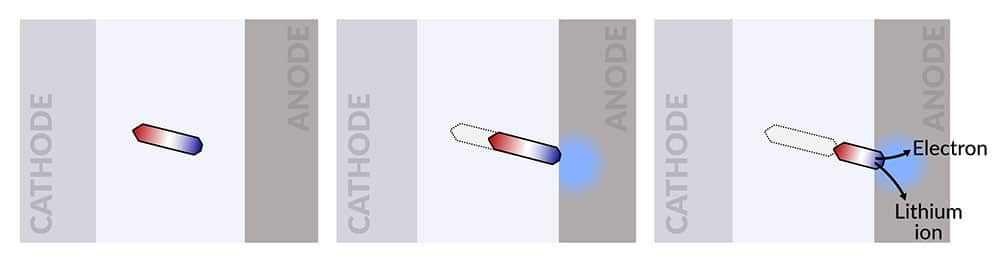
As lithium batteries cycle, they collect little islands of inactive lithium that are cut off from the electrodes, decreasing the battery’s capacity to store charge. But the research team discovered that they could make this “dead” lithium practically creep like a worm toward one of the electrodes until it reconnects, partially reversing the unwanted process.
When an island of inactivated lithium metal travels to a battery’s anode, or negative electrode, and reconnects, it comes back to life, contributing electrons to the battery’s current flow and lithium ions for storing charge until it’s needed. The island moves by adding lithium metal at one end and dissolving it at the other end. Researchers discovered that they could drive the island’s growth in the direction of the anode by adding a brief, high-current discharging step right after the battery charges. They found that reconnecting the island to the anode slowed the degradation of their test battery and increased its lifetime by nearly 30%.
The idea for the study was born when Yi Cui, a professor at Stanford and SLAC, who led the research, speculated that applying a voltage to a battery’s cathode and anode could make an isolated island of lithium physically move between the electrodes. His team has now confirmed the process with their experiments.
The team fabricated an optical cell with a lithium-nickel-manganese-cobalt-oxide (NMC) cathode, a lithium anode, and an isolated lithium island in between. This test device allowed them to track in real-time what happens inside a battery when in use.
They found that the isolated lithium island wasn’t really “dead” and responded to battery operations. When charging the cell, the island slowly moved towards the cathode; when discharging, it crept in the opposite direction.
The researchers validated the results with other test batteries and through computer simulations. They also demonstrated how isolated lithium could be recovered in a real battery by modifying the charging protocol.
“We found that we can move the detached lithium toward the anode during discharging, and these motions are faster under higher currents,” said Stanford postdoctoral fellow Fang Liu, the lead author of the study. “So we added a fast, high-current discharging step right after the battery charges, which moved the isolated lithium far enough to reconnect it with the anode. This reactivates the lithium so it can participate in the life of the battery.”
She also believes that their findings have wide implications for designing and developing more robust lithium-metal batteries.
