Fuel cells are a very promising technology for energy conversion, an area dominated by old technologies, such as the internal combustion engine and the gas turbine, which have to be replaced to allow decarbonization of the energy sector. Fuel cells offer high energy efficiency, extremely low pollutant emissions, low noise levels, and modularity.
Although fuel cells work just like batteries based on an electrochemical process, they don’t run down or require recharging. However, the potential advantages of fuel cells are offset by challenges that include cost, performance, and durability.
Now, researchers at Michigan Technological University took on those challenges, changing the conventional path of a fuel cell by creating an interface between the electrolyte and melted carbonate as an ultrafast channel for oxygen ion transfer. This enabled researchers to invent an entirely new type of fuel cell, a carbonate-superstructured solid fuel cell (CSSFC).
CSSFCs have a wide array of potential uses, from providing energy to operate fuel cell vehicles and home power generation to entire power stations. Because CSSFCs are fuel flexible, they offer higher durability and energy conversion efficiency at lower operating temperatures than other types of fuel cells.
Most fuel cells are powered by hydrogen – produced from hydrogen-containing compounds, mainly methane, via an expensive process. But the CSSFC developed by the researchers can directly use methane or other hydrocarbon fuels.
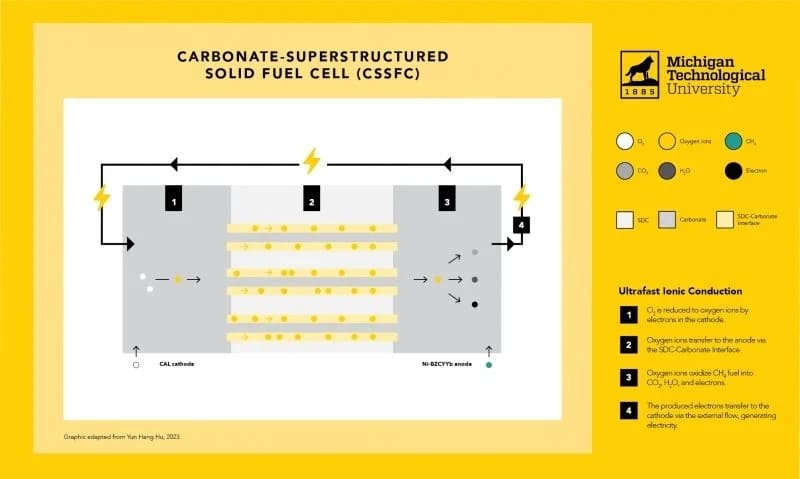
The new fuel cell’s electrochemical performance at lower operating temperatures offers several other advantages. “The operating temperature of a conventional solid oxide fuel cell is usually 800 degrees Celsius or higher because ion transfer in a solid electrolyte is very slow at a lower temperature,” said Yun Hang Hu, Charles, and Carroll McArthur, Endowed Professor of Materials Science and Engineering. “In contrast, the CSSFC’s superstructured electrolyte can provide a fast ion transfer at 550 degrees Celsius or lower — even as low as 470 degrees Celsius.”
The relatively low operating temperature offers high theoretical efficiency and lower cell fabrication costs. It is also potentially safer to operate than other solid fuel cells.
The lab tests on CSSFC also showed an unprecedentedly high open circuit voltage (OCV), which indicates no current leakage loss and high energy conversion efficiency. Hu estimates that CSSFC fuel efficiency could reach 60%, which is almost double the average fuel efficiency of a combustion engine ranges. The CSSFC’s higher fuel efficiency could lead to lower carbon dioxide emissions in vehicles.
“In our experiments, the CSSFC exhibited ultrahigh oxygen ionic conductivity at 550 degrees Celsius, achieving rapid oxidation of hydrocarbon fuel. This led to an unprecedented high open-circuit voltage of 1.041 volts and a very high peak power density of 215 milliwatts per square centimeter, along with excellent coking resistance using dry methane fuel,” said Hu.
He and his team will continue to look to new frontiers, including creating superstructured materials as a new platform for energy devices.
Journal reference:
- Hanrui Su, Wei Zhang, and Yun Hang Hu. Carbonate-superstructured solid fuel cells with hydrocarbon fuels. PNAS, 2022; DOI: 10.1073/pnas.220875011
