The fight against climate change is making the search for carbon-neutral energy sources increasingly urgent. Green hydrogen is a promising fuel in a carbon-neutral economy, and many efforts are currently being undertaken to produce hydrogen. One of the challenges is to store and transport the highly explosive gas in a safe and easy way.
Now, a team of microbiologists from Goethe University Frankfurt has succeeded in using bacteria for the controlled storage and release of hydrogen – an important step in the search for carbon-neutral energy sources in the interest of climate protection. Researchers found an enzyme in bacteria that live in the absence of air and bind hydrogen directly to CO2, in this way producing formic acid. The process is completely reversible – a basic requirement for hydrogen storage.
These acetogenic bacteria feed on carbon dioxide, which they metabolize to formic acid with the aid of hydrogen. Normally, this formic acid is just an intermediate product of their metabolism and is further digested into acetic acid and ethanol. But the team has adapted the bacteria in such a way that it is possible not only to stop this process at the formic acid stage but also to reverse it. The basic principle has been already patented since 2013.
“The measured rates of CO2 reduction to formic acid and back are the highest ever measured and many times greater than with other biological or chemical catalysts; in addition, and unlike chemical catalysts, the bacteria do not require rare metals or extreme conditions for the reaction, such as high temperatures and high pressures, but instead do the job at 30 °C and normal pressure,” reports Professor Volker Müller. The group now has a new success to report: the development of a biobattery for hydrogen storage with the help of the same bacteria.
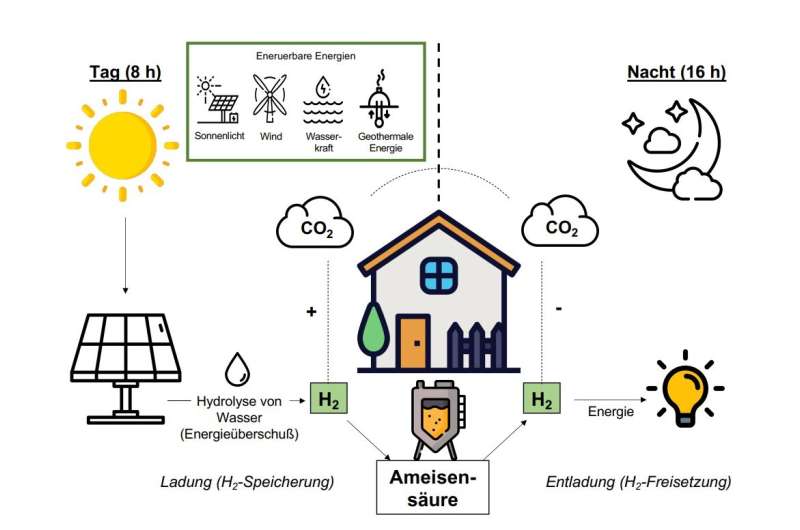
During the day, electricity is generated with the help of a photovoltaic unit, which then powers the hydrolysis of water. The bacteria bind the hydrogen produced in this way to CO2, resulting in the formation of formic acid. This reaction is fully reversible, and the direction of the reaction is steered solely by the concentration of the starting materials and end products. During the night, the hydrogen concentration in the bioreactor decreases, and the bacteria begin to release the hydrogen from the formic acid again. This hydrogen can then be used as an energy source.
To test their bacterial hydrogen storage system, the team fed the bacteria hydrogen for eight hours and then put them on a hydrogen diet during a 16-hour phase overnight. The bacteria then released all the hydrogen again. It was possible to eliminate the unwanted formation of acetic acid with the help of genetic engineering processes.
“The system ran extremely stably for at least two weeks,” explains Fabian Schwarz, who is pleased that this work has been accepted for publication in “Joule,” a prestigious journal for chemical and physical process engineering. “That biologists publish in this important journal is somewhat unusual,” says Schwarz.
According to researchers, the demonstrated process design can be considered as a future “bio-battery” for the reversible storage of electrons in the form of hydrogen in formic acid.
