The Medical robot specialist company CUREXO Corp. announced their independently developed spinal surgery robot ‘CUVIS-spine’ has obtained approval from the US Food and Drug Administration (FDA). This FDA license, followed by Korea (2019) and Europe CE (2020) certificate, is the third license that they have acquired, and now their product can be supplied to all countries.
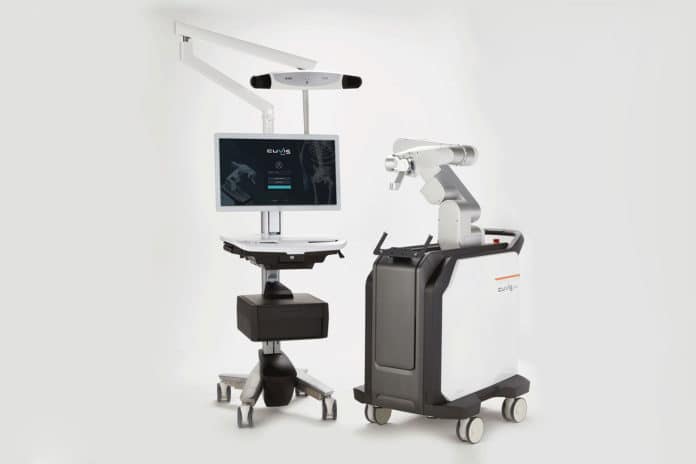
The ‘CUVIS-spine’ is a next-gen spinal surgery robot that guides the insertion of a pedicle screw according to the surgery plan. The surgical accuracy increases as it plans a surgery based on 2D C-arm or 3D CT images and corrects while monitoring the patient’s posture in real-time during the insertion of pedicle screws into patients.
The system uses a high-precision robot arm, wireless one-step navigation based on a real-time GPS sensor to provide precise, safe, and faster surgery compared to traditional manual surgery. It also minimizes the filming and reduces the radiation exposure of both patients and medical staff. This product can apply both 2-dimensional filming (C-arm) and 3-dimensional filming (O-arm), making it possible to target domestic and global markets.
Thanks to the Wireless One-step and the robot system, ‘CUVIS-spine’ allows multiple steps – such as Needle, K-wire, Dilation, Tapping – of traditional manual surgery to be performed with one surgery tool, and it reduces surgery duration greatly. It allows Minimally Invasive Surgery (MIS), which means that the patient’s recovery time will be quicker compared to manual surgery.
“The spinal surgery robot ‘CUVIS-spine’ that we have developed and manufactured with our technology has acquired FDA (US) ‘s licensing,” said Mr. Jae-Joon Lee, CEO of CUREXO. “This is the 3rd license that we have acquired, followed by Korea and Europe, and now we can sell our solution in all countries, including the US, which is the biggest medical market. CUREXO plans to focus on our medical robots sales, including ‘CUVIS-joint,’ ‘CUVIS-spine’ and ‘Morning Walk’ to not only the Korean market but also to advanced medical markets such as the US and Europe.”
Currently, two ‘CUVIS-spine’s are being used in Severance Hospital (Korea), and one has been exported to an Australian medical facility company. CUREXO also plans to expand its focus on global markets, including the US and Europe.